 Each element produces a unique emission spectrum.Įxamples of Various Spectra Gas Discharge Tubesĭischarge tubes are used to generate atomic emission spectra from gases.Ī discharge tube is a simple apparatus consisting of two metal electrodes connected to a high voltage potential difference. The absorption or emission spectrum would be unique for an element, and thus, can be used to identify elements.Įmission spectra of noble gases differ in the position and total number of emission lines. These streaks of light are called emission lines.Įlements have unique emission and absorption spectra.Ĭonsequently, no two elements will produce identical absorption and emission spectra. This produces an emission spectrum where only certain frequencies/wavelengths of light are present on a black background. The amount of energy released produces EMR of specific frequencies/wavelengths because it equals the difference between energy states. In a sample of hot gas where electrons are in their excited energy states due to the provision of heat as an energy source, these electrons can return to their ground states and release energy in the form of EMR. 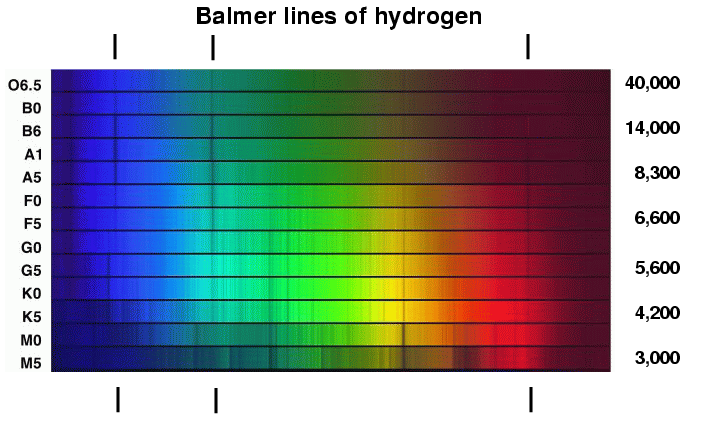
This type of spectrum is called an absorption spectrum.įor atoms of a particular element, there are usually more than one absorption line present because a ground state electron can be excited to different excited energy states by absorbing different amounts of energy (in the form of light). Therefore, absorption of light during electronic excitation causes there to be absent frequencies/wavelengths of light in the otherwise continuous spectrum. These absent lines are called absorption lines. When the same source of light is passed through atoms, ground state electrons can absorb specific amounts of energy to transition to higher energy states.ĭifferent energy of light corresponds to different frequencies/wavelengths. When white light is dispersed through a glass prism, it forms a continuous spectrum. White light consists of visible light of all wavelengths. Since atoms have multiple excitation states, there may be multiple electronic transitions, causing the emitted electromagnetic radiation to vary in energy and thus have different frequencies and wavelengths. When electrons return from their excited state to the ground state, energy is released in the form of electromagnetic radiation (EMR).ĭuring this process, electrons will release the same amount of energy as they absorbed (law of conservation of energy). The Hydrogen spectrum is an emission spectrum because the colored lines are separated by dark spaces, but the absorption spectrum has dark lines.If the energy provided does not exactly match this energy difference, it is does not absorbed by the electron.Įxcited electrons do not remain in their excited energy states permanently. Absorption spectrum is obtained when the white light if first passed through the substance and the transmitted light is analysed in the spectroscope. Emission spectrum is obtained when the radiation from the source is directly analysed in the spectroscope. The equation gives the calculation of the wave number ($\bar=\infty $. The Paschen series, Brackett series, and Pfund series belong to the infrared region.Īlthough a large number of lines are present in the hydrogen spectrum, Rydberg in 1890 gave a very simple theoretical equation for the calculation of the wavelength of these lines. The Balmer series belongs to the visible region. The Lyman series belongs to the ultraviolet region. The emission spectrum is similar except that in place of dark lines, there are colored lines with dark space in between. The names of different series are the Lyman series, Balmer series, Paschen series, Brackett series, and Pfund series. It is found to consist of a large number of lines that are grouped into different series. The emission spectrum of hydrogen or line spectrum of hydrogen is produced when hydrogen gas is taken in the discharge tube and the light emitted on passing electric discharge at low pressure is examined with a spectroscope. 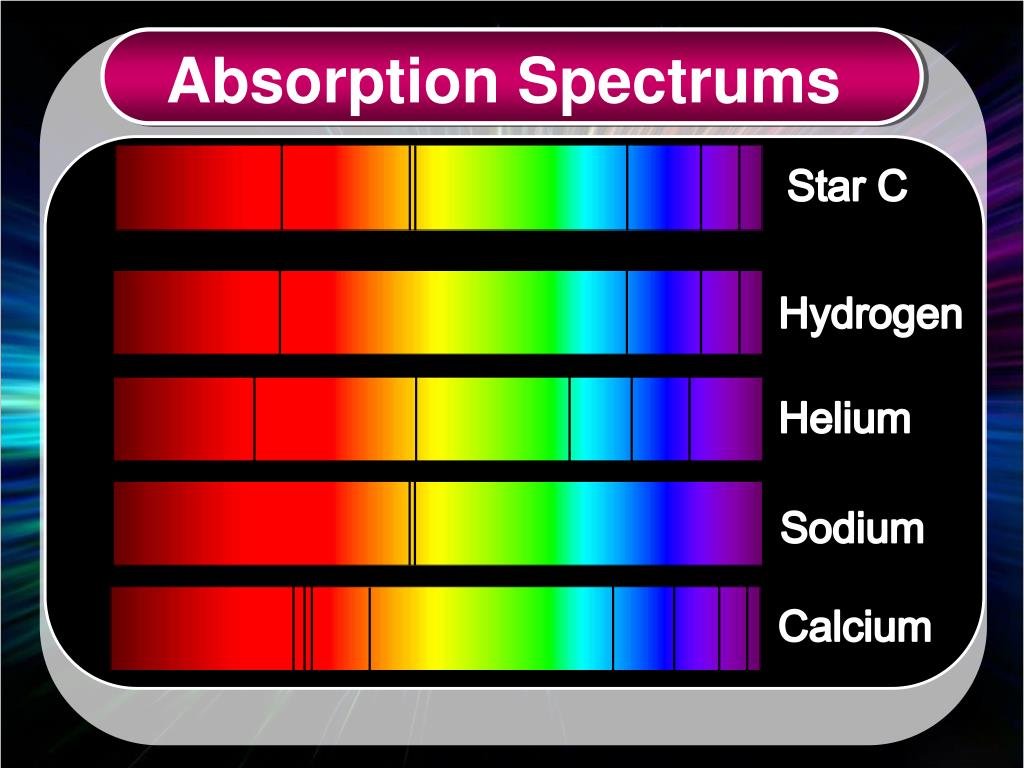
There are 5 series in the hydrogen spectrum, Lyman, Balmer, Paschen, Brackett, and Pfund series. Hint: The line spectrum of hydrogen is also known as the Emission spectrum of hydrogen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
